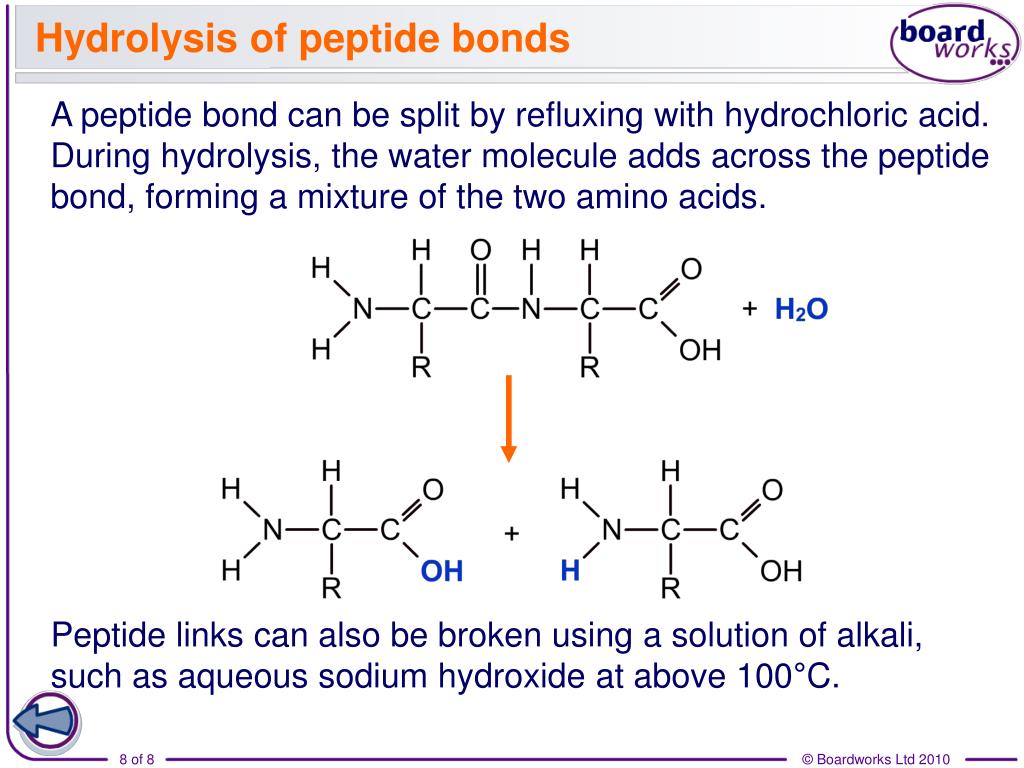
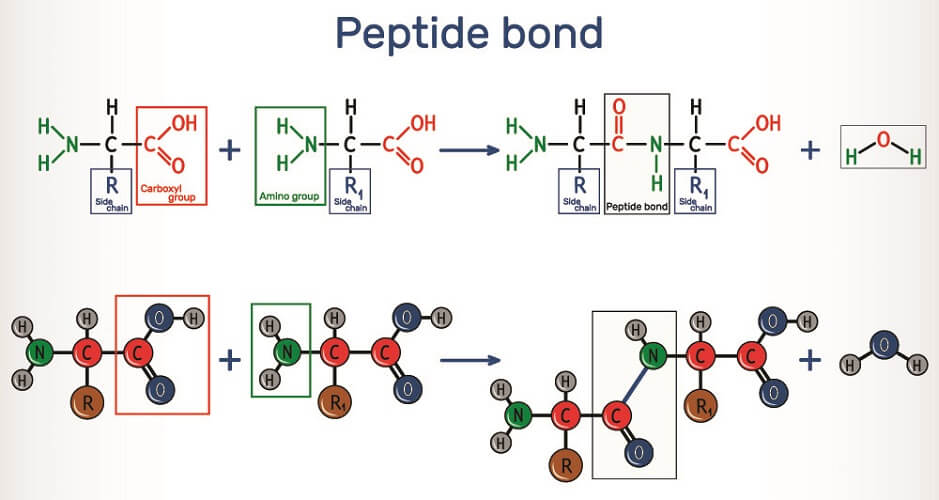
Are Peptide Bonds Formed By Hydrolysis - Peptide bond formation is an example of a condensation or dehydration reaction because it results in the removal of a water molecule (h2o);. They add an oh to the carbonyl carbon and an h to the amide nitrogen. The primary step in all protein hydrolysis reactions is peptide bond hydrolysis. Instead, they are created through dehydration synthesis, where amino acids combine and. A peptide bond is a chemical bond formed between two molecules when the carboxyl group of one molecule reacts with the amino group of the other. How do hydrolytic enzymes break peptide bonds? No, peptide bonds are not formed by hydrolysis.
The primary step in all protein hydrolysis reactions is peptide bond hydrolysis. They add an oh to the carbonyl carbon and an h to the amide nitrogen. Instead, they are created through dehydration synthesis, where amino acids combine and. A peptide bond is a chemical bond formed between two molecules when the carboxyl group of one molecule reacts with the amino group of the other. No, peptide bonds are not formed by hydrolysis. How do hydrolytic enzymes break peptide bonds? Peptide bond formation is an example of a condensation or dehydration reaction because it results in the removal of a water molecule (h2o);.
They add an oh to the carbonyl carbon and an h to the amide nitrogen. Peptide bond formation is an example of a condensation or dehydration reaction because it results in the removal of a water molecule (h2o);. How do hydrolytic enzymes break peptide bonds? No, peptide bonds are not formed by hydrolysis. The primary step in all protein hydrolysis reactions is peptide bond hydrolysis. A peptide bond is a chemical bond formed between two molecules when the carboxyl group of one molecule reacts with the amino group of the other. Instead, they are created through dehydration synthesis, where amino acids combine and.
Fundamentals of Anatomy & Physiology ppt download
Instead, they are created through dehydration synthesis, where amino acids combine and. They add an oh to the carbonyl carbon and an h to the amide nitrogen. How do hydrolytic enzymes break peptide bonds? No, peptide bonds are not formed by hydrolysis. Peptide bond formation is an example of a condensation or dehydration reaction because it results in the removal.
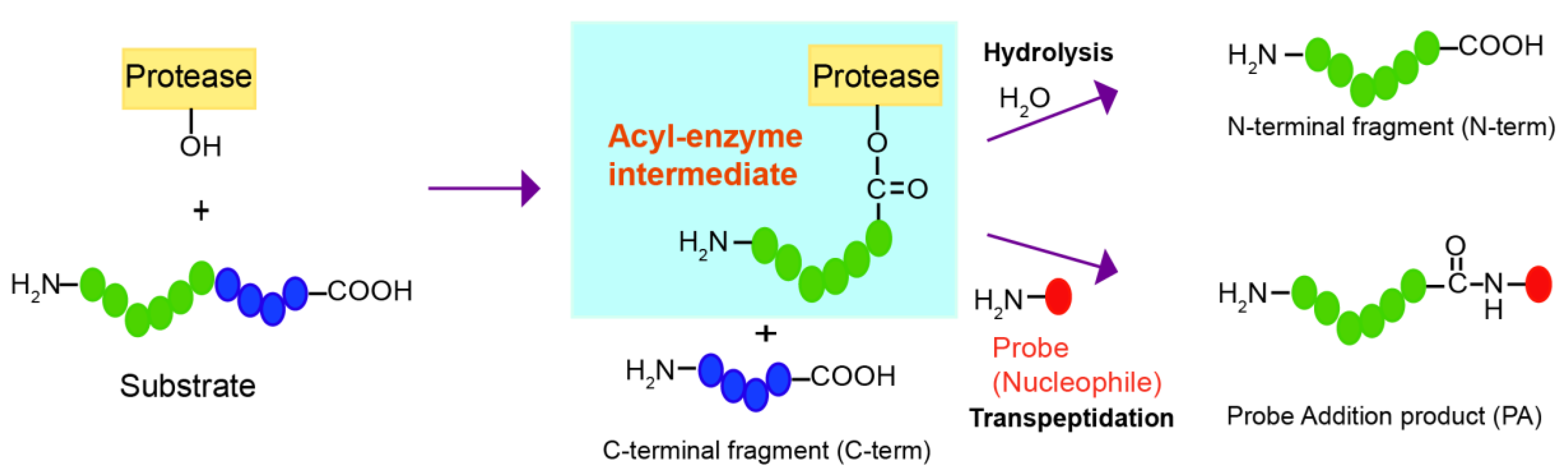
Peptide Bond Hydrolysis
No, peptide bonds are not formed by hydrolysis. A peptide bond is a chemical bond formed between two molecules when the carboxyl group of one molecule reacts with the amino group of the other. Peptide bond formation is an example of a condensation or dehydration reaction because it results in the removal of a water molecule (h2o);. The primary step.
PPT BioMolecules Organic compounds containing Carbon PowerPoint
Peptide bond formation is an example of a condensation or dehydration reaction because it results in the removal of a water molecule (h2o);. They add an oh to the carbonyl carbon and an h to the amide nitrogen. Instead, they are created through dehydration synthesis, where amino acids combine and. No, peptide bonds are not formed by hydrolysis. A peptide.
Biochem Block Handout 5 ppt download
The primary step in all protein hydrolysis reactions is peptide bond hydrolysis. A peptide bond is a chemical bond formed between two molecules when the carboxyl group of one molecule reacts with the amino group of the other. Instead, they are created through dehydration synthesis, where amino acids combine and. How do hydrolytic enzymes break peptide bonds? No, peptide bonds.
PPT Links to the current specifications PowerPoint Presentation, free
How do hydrolytic enzymes break peptide bonds? The primary step in all protein hydrolysis reactions is peptide bond hydrolysis. Peptide bond formation is an example of a condensation or dehydration reaction because it results in the removal of a water molecule (h2o);. A peptide bond is a chemical bond formed between two molecules when the carboxyl group of one molecule.
Peptide Bond Hydrolysis Mechanism PPT Summary ACP PPT PowerPoint
The primary step in all protein hydrolysis reactions is peptide bond hydrolysis. A peptide bond is a chemical bond formed between two molecules when the carboxyl group of one molecule reacts with the amino group of the other. Peptide bond formation is an example of a condensation or dehydration reaction because it results in the removal of a water molecule.
CZ5225 Modeling and Simulation in Biology Modeling and Simulation in
They add an oh to the carbonyl carbon and an h to the amide nitrogen. Instead, they are created through dehydration synthesis, where amino acids combine and. Peptide bond formation is an example of a condensation or dehydration reaction because it results in the removal of a water molecule (h2o);. No, peptide bonds are not formed by hydrolysis. How do.
Proteins (I). ppt download
Instead, they are created through dehydration synthesis, where amino acids combine and. The primary step in all protein hydrolysis reactions is peptide bond hydrolysis. No, peptide bonds are not formed by hydrolysis. Peptide bond formation is an example of a condensation or dehydration reaction because it results in the removal of a water molecule (h2o);. How do hydrolytic enzymes break.
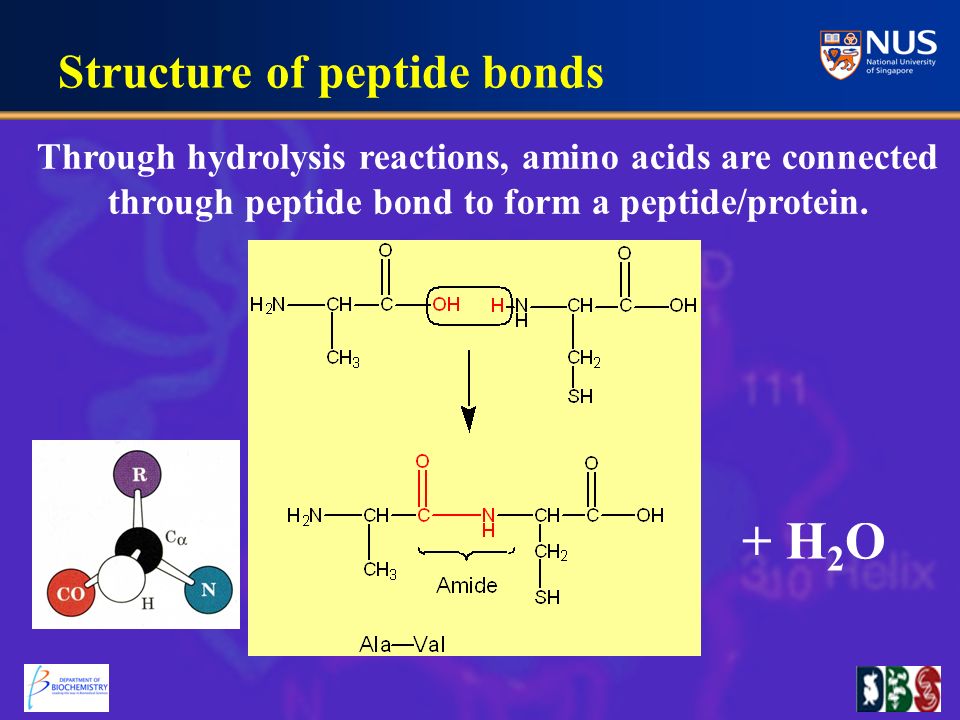
Proteins
The primary step in all protein hydrolysis reactions is peptide bond hydrolysis. Instead, they are created through dehydration synthesis, where amino acids combine and. They add an oh to the carbonyl carbon and an h to the amide nitrogen. A peptide bond is a chemical bond formed between two molecules when the carboxyl group of one molecule reacts with the.
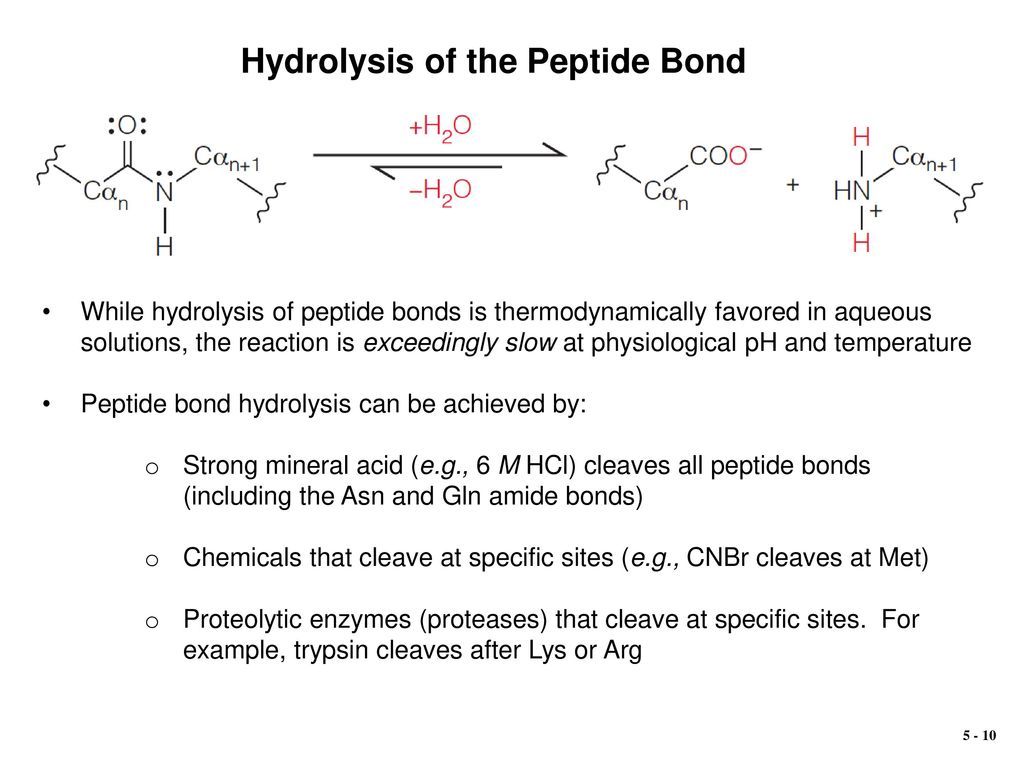
Hydrolysis Of Polypeptide
How do hydrolytic enzymes break peptide bonds? The primary step in all protein hydrolysis reactions is peptide bond hydrolysis. Instead, they are created through dehydration synthesis, where amino acids combine and. They add an oh to the carbonyl carbon and an h to the amide nitrogen. A peptide bond is a chemical bond formed between two molecules when the carboxyl.
How Do Hydrolytic Enzymes Break Peptide Bonds?
Instead, they are created through dehydration synthesis, where amino acids combine and. Peptide bond formation is an example of a condensation or dehydration reaction because it results in the removal of a water molecule (h2o);. The primary step in all protein hydrolysis reactions is peptide bond hydrolysis. A peptide bond is a chemical bond formed between two molecules when the carboxyl group of one molecule reacts with the amino group of the other.
No, Peptide Bonds Are Not Formed By Hydrolysis.
They add an oh to the carbonyl carbon and an h to the amide nitrogen.