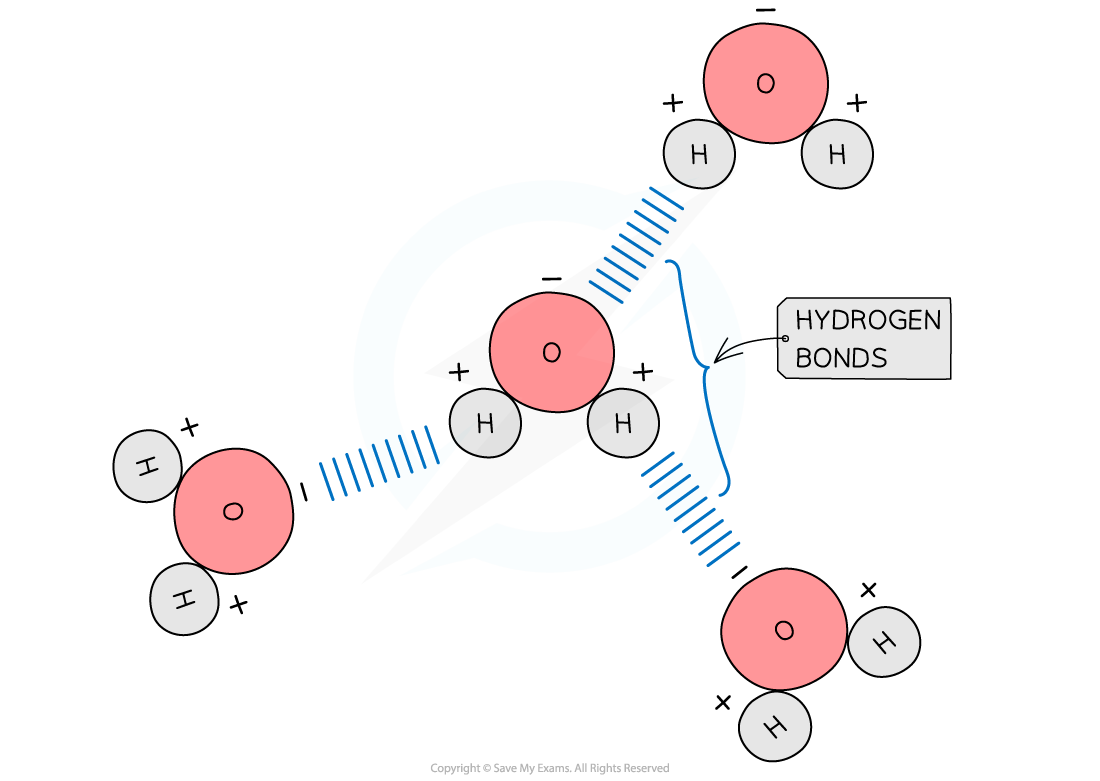
Hydrogen Bonds Form Between Adjacent Water Molecules Because The - Weak attractions (hydrogen bonds) form between adjacent water molecules, so that the slightly negative oxygen atom forms a weak bond. The intermolecular force that hold together adjacent water molecules are hydrogen bonds. Hydrogen bonds are the strongest type of. The weak bond that exists between water molecules is called a hydrogen bond. Hydrogen bonds can form between water molecules because the oxygen atom in water is slightly electronegative, pulling electron density towards. In a water molecule, h2o, each hydrogen atom forms a.
Weak attractions (hydrogen bonds) form between adjacent water molecules, so that the slightly negative oxygen atom forms a weak bond. Hydrogen bonds can form between water molecules because the oxygen atom in water is slightly electronegative, pulling electron density towards. The weak bond that exists between water molecules is called a hydrogen bond. In a water molecule, h2o, each hydrogen atom forms a. Hydrogen bonds are the strongest type of. The intermolecular force that hold together adjacent water molecules are hydrogen bonds.
Hydrogen bonds can form between water molecules because the oxygen atom in water is slightly electronegative, pulling electron density towards. In a water molecule, h2o, each hydrogen atom forms a. Weak attractions (hydrogen bonds) form between adjacent water molecules, so that the slightly negative oxygen atom forms a weak bond. The weak bond that exists between water molecules is called a hydrogen bond. The intermolecular force that hold together adjacent water molecules are hydrogen bonds. Hydrogen bonds are the strongest type of.
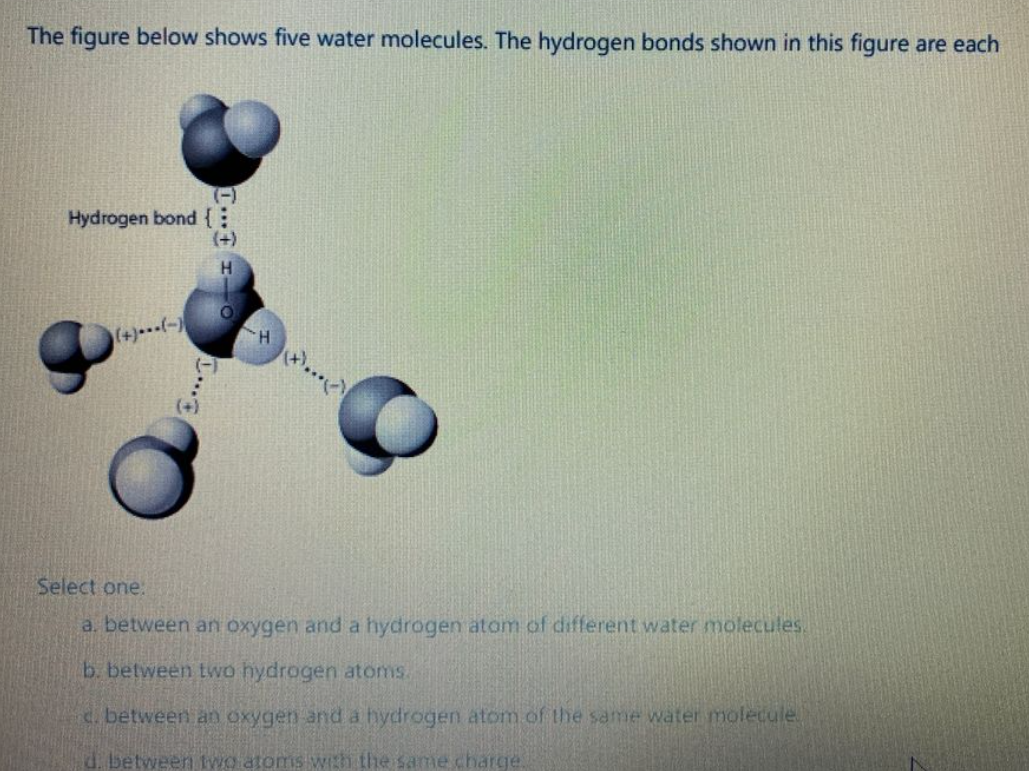
Solved The diagram shows hydrogen bonds between water molecules. Label
Weak attractions (hydrogen bonds) form between adjacent water molecules, so that the slightly negative oxygen atom forms a weak bond. Hydrogen bonds can form between water molecules because the oxygen atom in water is slightly electronegative, pulling electron density towards. The intermolecular force that hold together adjacent water molecules are hydrogen bonds. The weak bond that exists between water molecules.
IB DP Biology SL复习笔记2.1.3 Hydrogen Bonds翰林国际教育
Weak attractions (hydrogen bonds) form between adjacent water molecules, so that the slightly negative oxygen atom forms a weak bond. The intermolecular force that hold together adjacent water molecules are hydrogen bonds. In a water molecule, h2o, each hydrogen atom forms a. Hydrogen bonds are the strongest type of. The weak bond that exists between water molecules is called a.
Water is the medium of life. ppt download
Weak attractions (hydrogen bonds) form between adjacent water molecules, so that the slightly negative oxygen atom forms a weak bond. Hydrogen bonds are the strongest type of. Hydrogen bonds can form between water molecules because the oxygen atom in water is slightly electronegative, pulling electron density towards. The weak bond that exists between water molecules is called a hydrogen bond..
The Diagram Below Shows Hydrogen Bonding Between Water Molec
Weak attractions (hydrogen bonds) form between adjacent water molecules, so that the slightly negative oxygen atom forms a weak bond. The intermolecular force that hold together adjacent water molecules are hydrogen bonds. The weak bond that exists between water molecules is called a hydrogen bond. Hydrogen bonds are the strongest type of. Hydrogen bonds can form between water molecules because.
Diagram of hydrogen bonding between two water molecules Download
The weak bond that exists between water molecules is called a hydrogen bond. In a water molecule, h2o, each hydrogen atom forms a. The intermolecular force that hold together adjacent water molecules are hydrogen bonds. Hydrogen bonds are the strongest type of. Hydrogen bonds can form between water molecules because the oxygen atom in water is slightly electronegative, pulling electron.
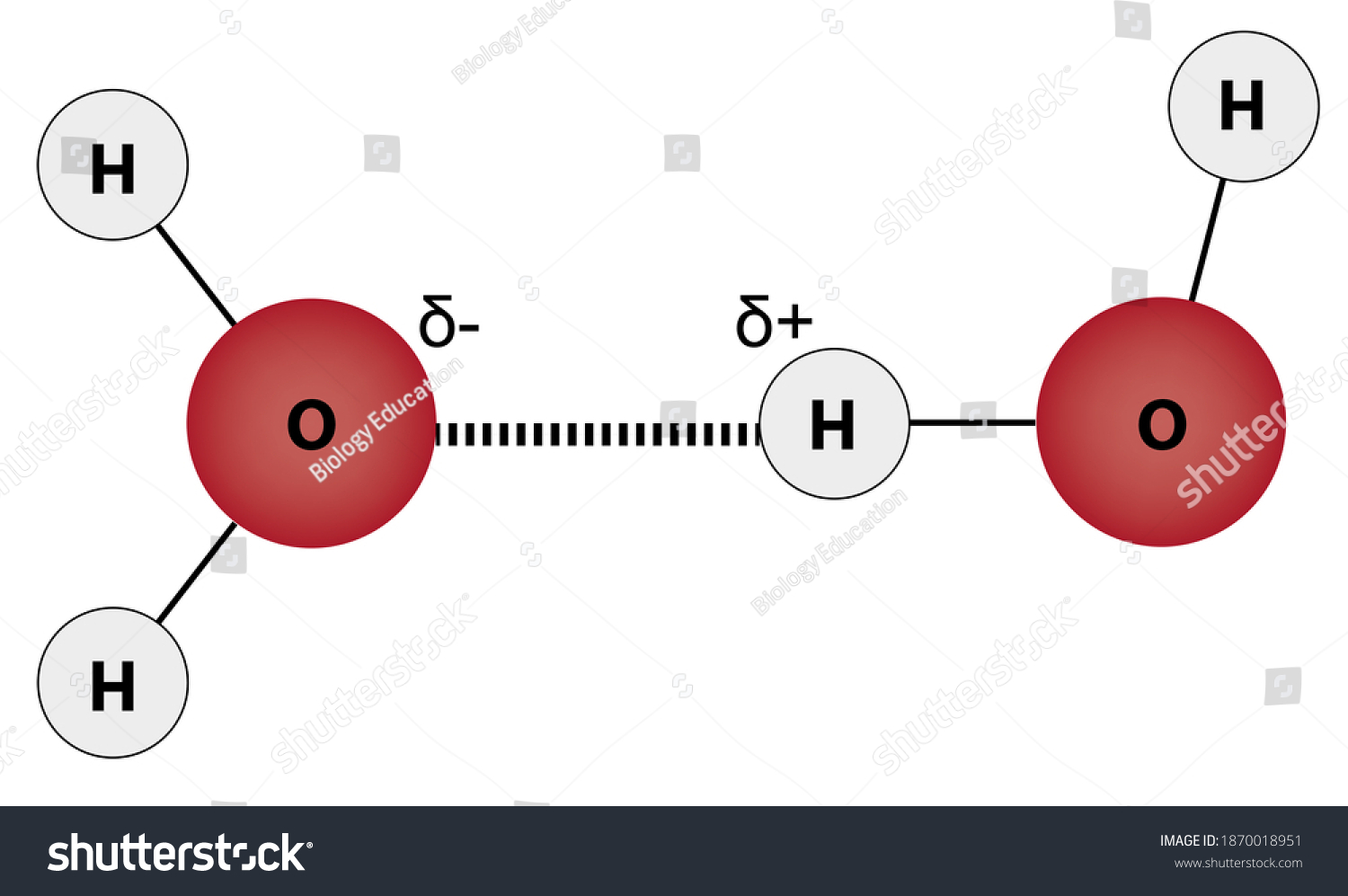
Water Molecules Joined By Hydrogen Bond Stock Illustration 1870018951
Weak attractions (hydrogen bonds) form between adjacent water molecules, so that the slightly negative oxygen atom forms a weak bond. Hydrogen bonds are the strongest type of. The intermolecular force that hold together adjacent water molecules are hydrogen bonds. In a water molecule, h2o, each hydrogen atom forms a. The weak bond that exists between water molecules is called a.
Basic Chemistry Water Acids, Bases and pH ppt download
Hydrogen bonds can form between water molecules because the oxygen atom in water is slightly electronegative, pulling electron density towards. Hydrogen bonds are the strongest type of. Weak attractions (hydrogen bonds) form between adjacent water molecules, so that the slightly negative oxygen atom forms a weak bond. The weak bond that exists between water molecules is called a hydrogen bond..
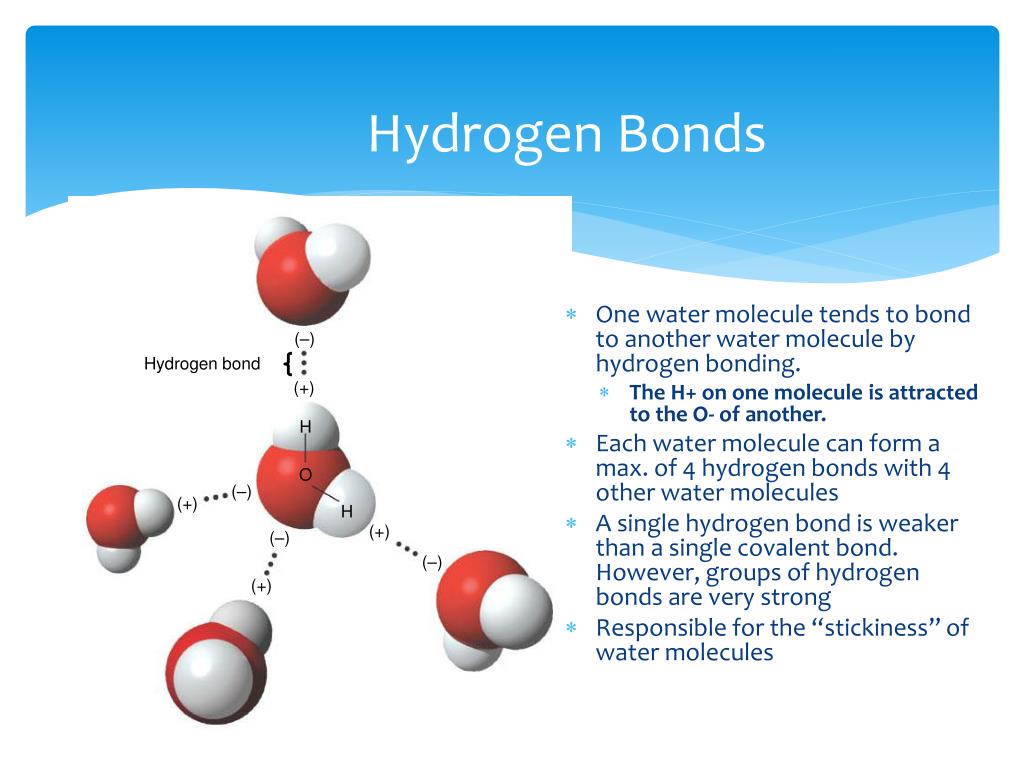
Hydrogen Bonds Between Water Molecules Diagram Science Onlin
Hydrogen bonds are the strongest type of. The intermolecular force that hold together adjacent water molecules are hydrogen bonds. The weak bond that exists between water molecules is called a hydrogen bond. Hydrogen bonds can form between water molecules because the oxygen atom in water is slightly electronegative, pulling electron density towards. In a water molecule, h2o, each hydrogen atom.
The diagram shows hydrogen bonds between water molecules. Label the
The weak bond that exists between water molecules is called a hydrogen bond. The intermolecular force that hold together adjacent water molecules are hydrogen bonds. Weak attractions (hydrogen bonds) form between adjacent water molecules, so that the slightly negative oxygen atom forms a weak bond. In a water molecule, h2o, each hydrogen atom forms a. Hydrogen bonds are the strongest.
The Diagram Shows Hydrogen Bonds Between Water Molecules. Wh
The weak bond that exists between water molecules is called a hydrogen bond. Weak attractions (hydrogen bonds) form between adjacent water molecules, so that the slightly negative oxygen atom forms a weak bond. In a water molecule, h2o, each hydrogen atom forms a. Hydrogen bonds can form between water molecules because the oxygen atom in water is slightly electronegative, pulling.
Weak Attractions (Hydrogen Bonds) Form Between Adjacent Water Molecules, So That The Slightly Negative Oxygen Atom Forms A Weak Bond.
Hydrogen bonds are the strongest type of. The intermolecular force that hold together adjacent water molecules are hydrogen bonds. The weak bond that exists between water molecules is called a hydrogen bond. In a water molecule, h2o, each hydrogen atom forms a.